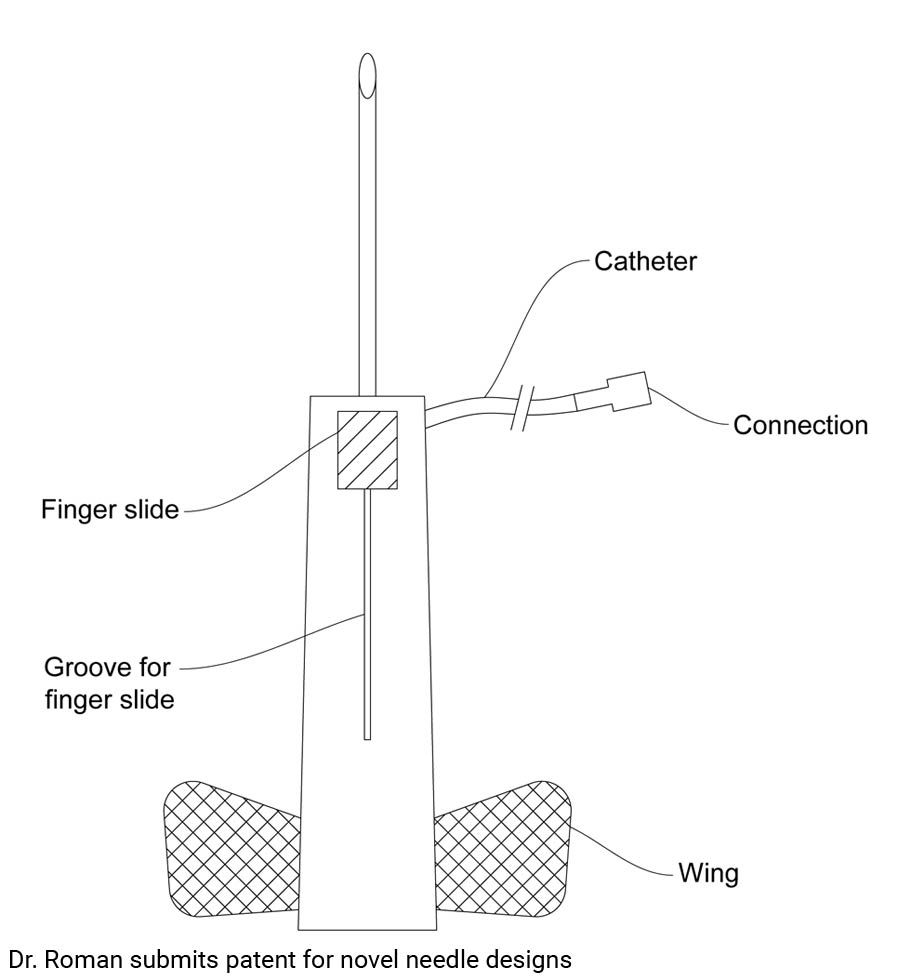
02 Dec Dr. Roman submits patent for novel needle designs
Reducing Bacterial Contamination of Cellular and Blood Products via Novel Needle Designs
Stephen Roman, M.D.
This is a brief summary describing the problem of bacterial contamination of cellular products and two novel methods designed to decrease its incidence.
The two main sources for cells in regenerative medicine and cell transplantation are peripheral vein blood draws and intra-osseous marrow extraction. The reported rate of contamination of the various types of stem cells ranges from 0.2% to more than 24%, averaging about 3% (Jacobs MR) (Kamble R) (Namdaroglu S). For HPCs sourced from peripheral blood (PB), the incidence of contamination is close to 2.1%. Rates of bacterial contamination of bone marrow-derived HPCs, are generally higher than PB-HPCs, reaching levels >3.5% (Honohan A).
Despite careful attention to sterile procedures, low-level contamination of hematopoietic stem cell components can occur. Bacteria from the skin flora (coagulase-negative Staphylococcus, Propionibacterium and Corynebacterium species) are the contaminants in approximately 90% of cases (de Medeiros CRFL).
The pathogenicity of skin flora is a complex topic. Let’s take for example Cutibacterium Acnes. Cutibacterium (formerly known as Propionibacterium) species are nonsporulating, gram-positive anaerobic bacilli. P. acnes colonizes primarily sebaceous glands of hair follicles of human skin, but it may also be found in the mouth, nares, genitourinary tract, and large intestine. (Ackermann) In 2004, the whole genome of C.acnes was sequenced by Bruggemann et al. (Bruggemann H). They concluded the genome sequence clearly reveals many proteins involved in the ability of P. acnes to colonize and reside in human skin sites as well as a pronounced potential to survive a spectrum of environments. This capacity helps to explain the ubiquity of C. acnes and also its potential hazards, for example, the public health problems associated with Blood Bank contaminations. Although primarily recognized for its role in acne, C. acnes is an opportunistic pathogen, causing a range of postoperative and device-related infections. These include infections of the bones and joints, mouth, eye and brain. Device-related infections include those of joint prostheses, shunts and prosthetic heart valves. C. acnes may play a role in other conditions, including inflammation of the prostate leading to cancer, SAPHO (synovitis, acne, pustulosis, hyperostosis, osteitis) syndrome, sarcoidosis and sciatica (Perry A). McDowell et al. (McDowell A) demonstrated that some types of C. acnes appear to be commensural, while others have been found in human infections with some resistant to multiple antibiotics. C. acnes is able to invade mesenchymal signaling cells (MSCs) inducing the potential transition of commensal C. acnes to an opportunistic pathogen in implant-associated infections (i.e. by increasing biofilm formation and resistance of macrophage phagocytosis) (Dubus M).
Contamination of cell cultures is not only frustrating, but is also very expensive both in time and loss of materials. (Vierck JL).
Contamination in a case of bone marrow transplantation is usually symptom free, however this may result in bacteremia with febrile illness (Stroncek DF) (Webb IJ) (Lazarus HM) (Namdaroglu S) as well as in septic shock (de Medeiros CRFL).
Infections resulting from skin commensals can be very dangerous in neutropenic patients and resistance has been increasing ie. among S. epidermidis (Lowder JN).
In the growing field of regenerative medicine, blood products such as platelet rich plasma (PRP), bone marrow and adipose tissue are now being used on a regular basis. Contamination of these cellular products is possibly the cause of severe complications. Two cases of spondylodiscitis (Subach BR) (Beatty NR) have been reported after intradiscal treatment with autologous cellular products. These infections may have serious sequelae, some requiring prolonged hospitalization, and some necessitating lumbar fusion surgery.
With the growing numbers of patients receiving cellular transplantation treatments as well as regenerative procedures, some involving culturing of cells prior to administration, it is more important than ever to prevent bacterial contamination of these products.
Up to 20% of skin bacteria are in the deeper layers of the skin and the pilo-sebaceous units, and these areas are untouched by antiseptics (Selwyn S) (Caldeira D). Additionally, contamination of the surface of the skin can occur immediately after disinfection, or an antiseptic may insufficiently eradicate certain pathogens from the needle site (Zubrod CJ) (Dumville JC). Therefore, despite adherence to the most stringent aseptic skin preparation techniques, a core of skin tissue with microbial flora may be introduced into a phlebotomy needle, infecting a cellular sample. In a cadaver study, it was noted that needle coring occurs often and could be a possible cause of post-injection septic arthritis (Xu C).
I am a specialist in the fields of regenerative medicine, internal medicine, pain management, physical medicine and rehabilitation , and headache management. I have performed in depth research on the needles in use and found that my two inventions are an improvement in the art, and that the ideas would be useful tools in medical applications. There was a comprehensive study of the prior art of issued and pending patents, and it was determined that the ideas I have outlined are novel, and in our opinion patentable. Based on that research, I have patent pending status the United States for these novel designs.
I have designed a novel phlebotomy needle to decrease the incidence of contamination of blood samples. I have also designed a novel bone marrow biopsy needle to not only decrease the incidence of contamination, but to also increase ease of operation and to avoid possible fracture or other complications (Gladden K) (Bain BJ). I have patent pending status and I am looking for a strategic partnership to bring these improved needles to market.
References:
- Achermann Y, Goldstein EJC, Coenye T, Shirtliff ME. Proprionibacterium acnes: from commensal to opportunistic biofilm-associated implant pathogen. Clinical Microbiology Reviews. 2014;27(3):419-40.
- Bain BJ. Morbidity associated with bone marrow aspiration and trephine biopsy—a review of UK data for 2004. Haematologica 2006;91:1293.
- Beatty NR, Lutz C, Boachie-Adjei K, Leynes TA, Lutz C, Lutz G. Spondylodiscitis due to Cutibacterium acnes following lumbosacral intradiscal biologic therapy: a case report. Regenerative Medicine 2019;Aug 19.
- Bruggemann H, Henne A, Hoster F, Liesegang H, Weizer A, Strittmatter A, Jujer S, Durre P, Gottschalk G. The Complete Genome Sequence of Propionibacterium Acnes, a Commensal of Human Skin. Science 2004;305(5684):671-673.
- Caldeira D, David C, Sampaio C. Skin antiseptics in venous puncture-site disinfection for prevention of blood culture contamination: systematic review with meta-analysis. Journal of Hospital Infection 2011;77:223e232.
- de Medeiros CRFL, Friedrich ML, Pereira N, Cunha CA, Pasquini R: Unrelated bone marrow as a source of Bacillus cereus causing septic shock. Bone Marrow Transplant 2000; 26:1259–1262.
- Dubus M, Varin J, Papa S, et al. Interaction of Cutibacterium acnes with human bone marrow derived mesenchymal stem cells: a step toward understanding bone implant associated infection development. Acta Biomaterialia 2020;104:124–134.
- Dumville JC, McFarlane E, Edwards P, Lipp A, Holmes A, Liu Z. Preoperative skin antiseptics for preventing surgical wound infections after clean surgery (Review). Cochrane Database Syst Rev, 2015; (4): CD003949.
- Gladden K, Spill GR. Iliac Fracture After a Bone Marrow Biopsy. PM&R, 2011, Vol.3(12), pp.1150-1152.
- Honohan A, Olthuis H, Bernards AT et al. Microbial contamination of cord blood stem cells. Vox Sang 2002;82:32-38.
- Jacobs MR, Good CE, Fox RM, et al.: Microbial contamination of hematopoietic progenitor and other regenerative cells used in transplantation and regenerative medicine. Transfusion 2013; 53:2690–2696.
- Kamble R, Pant S, Selby GB, et al.: Microbial contamination of hematopoietic progenitor cell grafts-incidence, clinical outcome, and cost-effectiveness: an analysis of 735 grafts. Transfusion 2005; 45:874–878.
- Lazarus HM, Magalhaes-Silverman M, Fox RM, et al. Contamination during in vitro processing of bone marrow for transplantation: clinical significance. Bone Marrow Transplant 1991;7:241-246.
- McDowell A, Barnard E, Nagy E, Gao A, Tomida S, Li H, Eady A, Cove J, Nord CE, Partick S. An Expanded Multilocus Sequence Typing Scheme for Propionibacterim acnes: Investigation of ‘Pathogenic’, ‘Commensal’ and Antibiotic Resistant Strains. PLoS ONE 2012;7(7): e41480. doi:10.1371/journal.pone.0041480.
- Namdaroglu S, Tekgunduz E, Bozdag SC, et al.: Microbial contamination of hematopoietic progenitor cell products. Transfus Apher Sci 2013; 48:403– 406.
- Perry A, Lambert P. Propionibacterium acnes: infection beyond the skin, Expert Review of Anti-infective Therapy 2011;9(12):1149-1156. DOI: 10.1586/eri.11.137
- Selwyn S, Ellis H. Skin Bacteria and Skin Disinfection Reconsidered. British Medical Journal 1972;.1(5793);136-140.
- Stroncek DF, Fautsch SK, Lasky LC, et al. Adverse reactions in patients transfused with cryopreserved marrow. Transfusion 1991;31:521-6.
Subach BR, Copay AG, Marcus MM, Schuler TC, DeWolfe DS. Epidural abscess and cauda equina syndrome after percutaneous intradiscal therapy in degenerative lumbar disc disease. The Spine Journal 2012;12:e1-e4. - Vierck JL, Byrne K, Mir PS, Dodson MV. Ten commandments for preventing contamination of primary cell
cultures. Methods in Cell Science 2000;22:33–41. - Webb IJ, Coral FS, Andersen JW et al. Sources and sequelae of bacterial contamination of hematopoietic stem cell components: implications for the safety of hematotherapy and graft engineering. Transfusion 1996; 36: 782–788.
- Xu C, Peng H, Chai W, Li R, Chen J. Inadvertent Introduction of Tissue Coring Into Joints During Arthrocentesis: An Experimental Study. Med Sci Monit. 2017;23:3571-3577.
- Yayac M, Schiavo P, Roman S. Chronic Low Back Pain With Modic Changes Treated with Doxycycline. Presented at the Interventional Orthobiologics Foundation 2020 Annual meeting. https://interventionalorthobiologics.org/wp-content/uploads/2020/04/Yayac-Abstract.pdf
- Zubrod CJ, Farnsworth KD, Oaks JL. Evaluation of Arthrocentesis Site Bacterial Flora before and after 4 Methods of Preparation in Horses with and without Evidence of Skin Contamination. Veterinary Surgery 2004;33:525–530.



Sorry, the comment form is closed at this time.